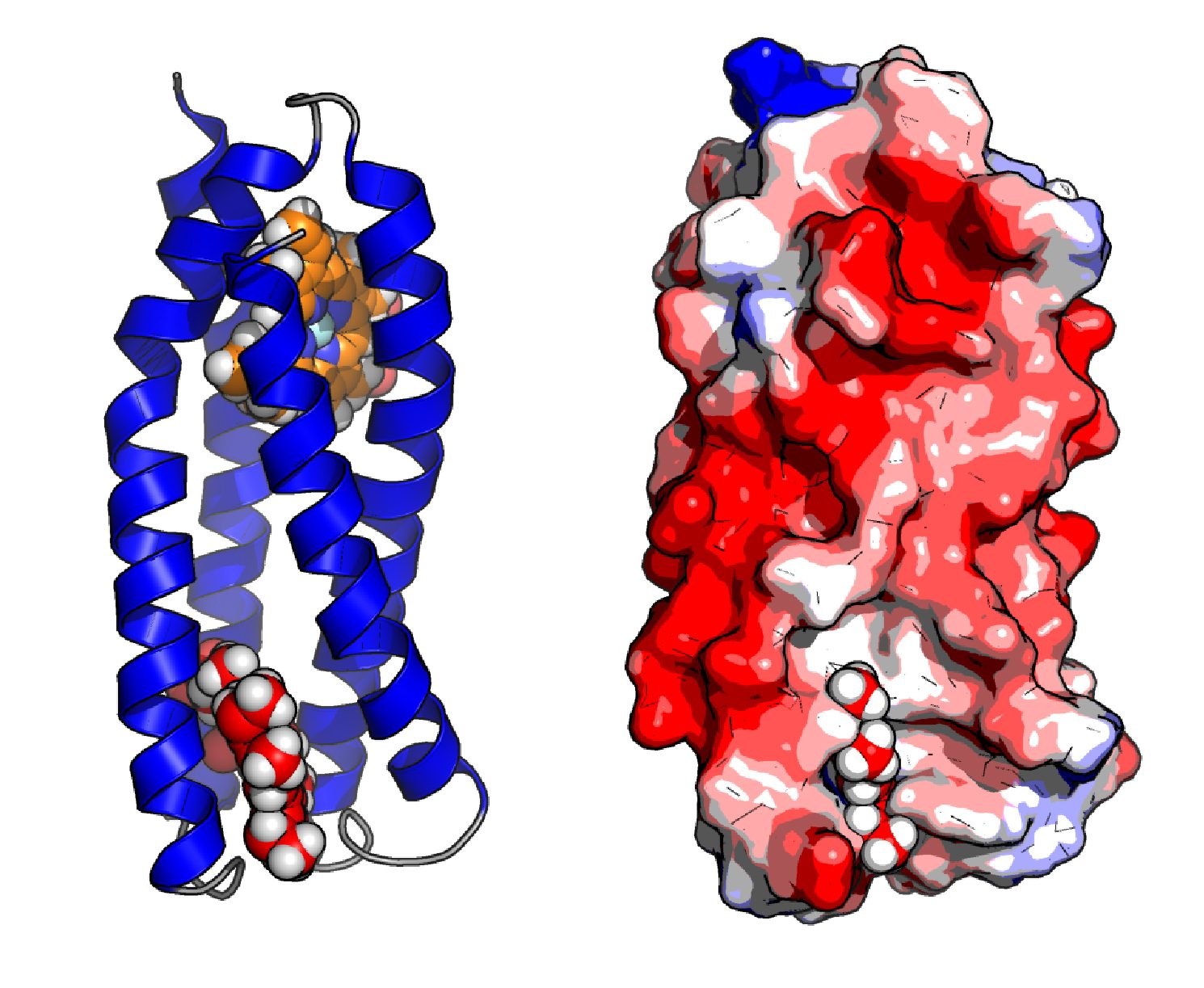
My interests lie in protein design, aiming to engineer new artificial enzymes. The key strategy in the design of such enzymes is to combine simple de novo protein scaffolds, designed to form helical bundle structures, with the diverse chemistry offered by redox cofactors such as heme groups. The goal is to target these enzymes towards the catalysis of specific reactions, and eventually integrate them into larger biological systems, contributing to the ever-expanding field of synthetic biology.

Supervised by both Dr. Ross Anderson in Biochemistry and Prof. Adrian Mulholland in Chemistry, I share my time between the two departments, combining computational design and characterisation of proteins with experimental investigation into new designs. My interests in computational chemistry range from classical MD to assess the design of structurally stable proteins, to QM/MM calculations to study the reactivity of newly designed enzymes.
A current focus is the design of structurally characterised proteins through crystallography and NMR, as previously designed proteins tend to be highly flexible and incompatible with structural biology. Computational design of proteins to develop tightly packed, organised core structures may aid in our structural understanding of such de novo protein designs.
My PhD is funded by the BBSRC as part of the SWBio Doctoral Training Partnership.